Background
Patients undergoing long-term glucocorticoid therapy are administered additional glucocorticoids
before minor dental procedures, although this is not supported by evidence. The authors
designed this study to validate the hypothesis that routine blanket glucocorticoid
supplementation is unnecessary during minor oral surgical procedures under local anesthesia.
Methods
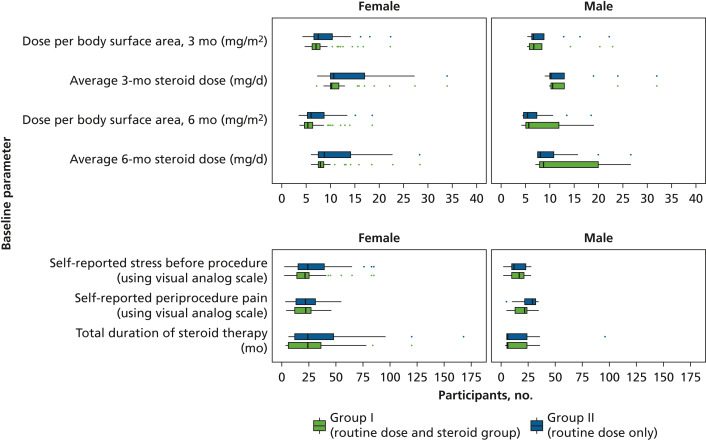
The authors recruited 270 patients into 3 groups (1:1:1 allocation) from the dental
outpatient department. Primary outcomes were changes in hemodynamic parameters and
frequency of adverse events among the 3 groups. The secondary outcome was the association
of preprocedural stress and procedural pain with periprocedural adverse events in
the long-term glucocorticoid therapy group (groups I and II).
Results
No clinically relevant changes in hemodynamic parameters among the 3 groups were found.
The authors also found low periprocedural adverse events in all 3 groups combined
(n = 1), so they did not explore the secondary outcomes further.
Conclusions
Among patients undergoing long-term glucocorticoid therapy for indications other than
primary adrenal insufficiency, elective minor oral surgical procedures can be performed
safely with only their daily dose of glucocorticoid when their medical conditions
are optimized. Routine additional glucocorticoid supplementation appears unnecessary.
The results of the study also revealed opportunities for value addition by means of
integrating oral health care with medical follow-up for patients with multiple co-occurring
medical conditions.
Practical Implications
Routine blanket glucocorticoid supplementation among patients taking a long-term glucocorticoid
for indications other than primary adrenal insufficiency appears unnecessary before
minor oral surgical procedures under local anesthesia. This clinical trial was registered
at Clinical Trial Registry-India. The registration number is CTRI/2017/02/007779.