MSCs isolation has been attempted from various tissues to date, including the bone marrow15, adipose tissue (ADSCs)16,17, umbilical cord blood (UCB-MSCs)18,19, and periodontal tissue. After hDPSCs isolation1 and SHED isolation2, isolation of stem cells from the apical papilla4,20 and periodontal ligament21 was achieved. The success rate of the isolation of these cells is essential for their clinical applications. The success rate of BMMSCs and ADSCs was reported to be 100%22, whereas the success rate of UCB-MSCs is only 29%. However, this could be increased by 63% by precoating the culture dish with foetal calf serum taking into account only units of optical quality23. The success rate of MSC isolation from cryopreserved UCB is below 57%24. However, there are few reports related to the isolation rate of hDPSCs. Eubanks et al.25 reported that the success rate of hDPSCs isolation using FBS was 89%, and Lee et al.26 reported that cryopreservation of teeth negatively affected the isolation of hDPSCs, with a success rate of only 73%. Similarly, we found a 70% success rate of hDPSCs using FBS. However, this is the first report of the SHED isolation rate by using FBS, demonstrating improved success at 81.8%. Although these rates are relatively high, clinical application requires a rate as close to 100% as possible. The success rate of SHED may depend on which tooth is selected. Selection criteria need to be identified to improve the success rate.
Moreover, serum-free medium (SFM) or autoserum is needed to isolate the MSCs because long-term culture using medium containing FBS may result in the differentiation and malignant transformation of the cells. In addition, the use of FBS is associated with increased risks of infections of prions and pathogenic viruses. Therefore, the use of SFM is strongly recommended for clinical applications27,28,29,30. However, Karbanove et al.31 reported that SFM decreased the proliferative and differentiation ability of hDPSCs compared to culture with FBS. By contrast, Hirata et al.30 reported no significant difference in the survival rate of SHED culture with and without SFM, although SFM increased the proliferation ability. In any case, there is no doubt that more reliable research without using FBS or SFM is needed in the near future.
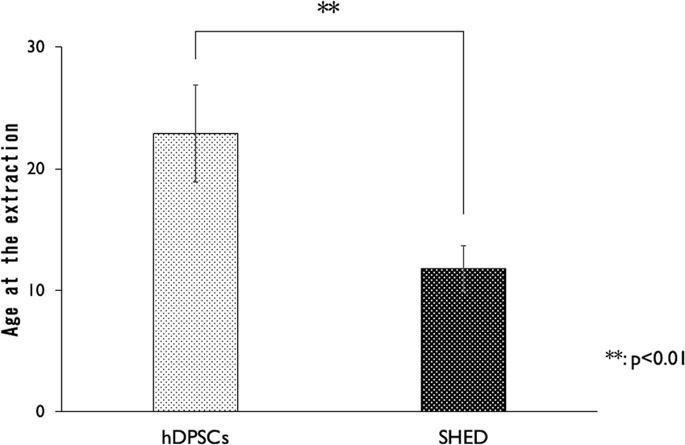
In our study, the patients in the SHED group were clearly younger than those in the hDPSCs group. This difference is expected because the reason for extraction in the SHED group was the eruption of permanent teeth and prolonged retention of deciduous teeth, whereas the teeth in the hDPSCs group were extracted for orthodontic treatments such as for the extraction of premolars and wisdom teeth. There was no significant difference in the success of isolation from both permanent and deciduous teeth. In SHED, because the success rate of cell isolation was high, the sample size of the failure group was very small. Therefore, despite significance using the Mann–Whitney U test, the results may not be statistically reliable. Furthermore, according to the condition of the teeth in the failure group, not only the age of patients when teeth were obtained, but also mechanical stress and other factors, might be associated with the success rate of the isolation.
The isolation of SHED was confirmed at a significantly earlier time during culture than that of hDPSCs. However, the standard deviation of the period from plating to confirmation was 4.9 days in hDPSCs and 5.5 days in SHED, indicating substantial individual variation.
To our knowledge, this is also the first report on the effect of the remaining root length ratio after physiological root absorption at the time of harvesting cells on the isolation success. In this study, the morphology of the tooth root and the number of roots of the tooth differed between the anterior teeth and the molar teeth. Therefore, these teeth were analysed separately. Furthermore, since the panoramic radiographs were not taken just before the extraction for some of the patients, the degree of absorption of the tooth root was calculated based on the norm values reported by Black et al.14. The remaining root length ratio of the successful group of anterior primary teeth was 72.4% (n = 13). However, this included one case in which the remaining root length ratios were only 30.1% and 43.2%, which could have influenced this result. Although the root length ratio of molars was only 61.4%, there were only four such teeth included in this group. However, the shortest root length ratio observed in this study was in a molar (28.6%) for which stem cell isolation failed, while another molar showed the greatest root length ratio overall (92.4%), although the following permanent tooth was a congenital missing tooth. Furthermore, since attrition was observed, and the patient was 21 years old, there was a possibility that the tooth had undergone long-term mechanical stress. Moreover, at the time of obtaining of the pulp tissue, stenosis of the root canal was observed, and the amount of collected pulp tissue was also very small. These facts suggest that the teeth most suitable for SHED isolation are those with only mild progression of root absorption and without mechanical stimulation. In addition, the SHED success rate is also strongly related to the root condition of the obtained deciduous teeth. Furthermore, it is necessary to consider not only the length of the remaining root but also the mechanical stress to the teeth, among other factors.
Accumulating evidence points to the potential of dental pulp as an accessible and promising cell source for bone regeneration7,8,9,10,11,12,13. In the near future, SHED and hDPSCs may become some of the best candidates for tissue regeneration. Therefore, more extensive studies and data on optimal methods for obtaining these cells will be of great importance to provide a safe and reliable regenerative medicine strategy.